Xenon (Xe) and Krypton (Kr) are not “nice-to-have” commodities anymore—they’re strategic inputs for semiconductor fabs, aerospace propulsion programs, and advanced manufacturing. The challenge is that these noble gases exist at extremely low concentrations in air and are typically obtained only as trace by-products of Air Separation Units (ASUs). That reality drives two painful outcomes for end users: scarcity and price volatility.
The engineering answer is rare gas recovery: capturing “crude” Kr/Xe-rich streams and upgrading them—reliably and repeatedly—into ultra-high purity (UHP) product grades such as 6N (99.9999%).
At Fortune Gas, rare gas recovery is treated as a full process discipline—combining cryogenic engineering, purification know-how, and robust equipment design to turn bypass/purge streams into high-value assets.
In high-purity gas specifications, the “N” notation indicates the number of nines in the purity level:
6N = 99.9999% purity
That corresponds to ~1 ppm total impurities (and in many real UHP specs, key impurities are controlled down to ppb levels depending on the application and analytical method).
Why this matters: in semiconductor and propulsion contexts, the impurities you worry about are often not “bulk” contaminants, but trace poisons that can impact yield, optics, or thruster stability—especially:
Moisture (H₂O)
Oxygen (O₂)
Nitrogen (N₂)
Hydrocarbons (THC)
Engineers evaluating a recovery project should treat “6N” as more than a marketing label: it’s the outcome of tight process control, materials compatibility at cryogenic temperatures, and credible trace impurity analysis.
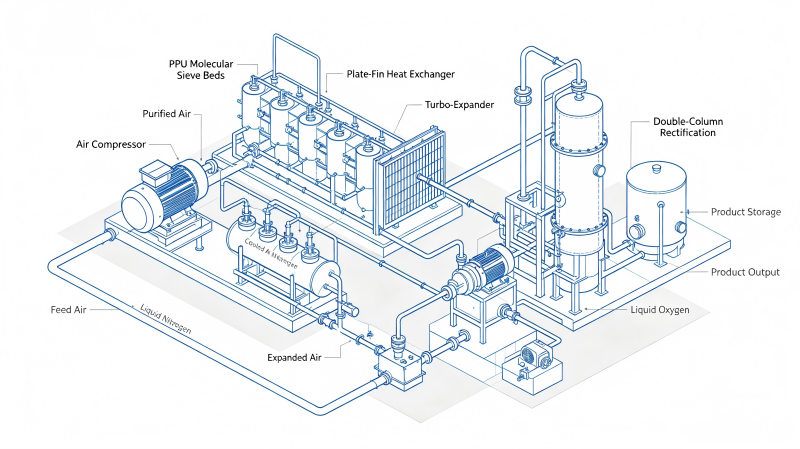
Rare gas recovery for Kr/Xe is typically integrated with, or downstream of, an ASU. In most industrial designs, Kr and Xe are first concentrated together in an oxygen-enriched stream and then purified using a sequence of catalytic, adsorption, and cryogenic separation steps.
In a cryogenic ASU, krypton and xenon tend to concentrate in certain oxygen-rich streams. Plants often pull a side stream (or purge) that contains:
O₂ as the dominant component
Trace to low-percentage levels of Kr/Xe (plus other minor components)
This is the starting point for rare gas recovery: you’re not “making” xenon/krypton—you’re concentrating and upgrading what the ASU already “collects” naturally.
Even tiny amounts of hydrocarbons (e.g., methane) or hydrogen in oxygen-enriched feeds can be problematic for both safety and purification performance. A catalytic oxidation stage converts these into species that are easier to remove downstream (typically CO₂ and H₂O). This “front-end cleanup” is commonly referenced in Kr/Xe recovery approaches tied to ASU oxygen-enriched streams.
Next, a pre-purification unit (often molecular sieve beds) is used to remove:
Moisture (H₂O)
CO₂
sometimes other strongly adsorbing contaminants
This protects cryogenic equipment from freeze-out/plugging and stabilizes downstream distillation performance.
The “heart” of high-grade Kr/Xe production is usually cryogenic distillation—a fractional distillation column system supported by high-performance heat exchange and careful thermal integration.
Why distillation works here: krypton and xenon have different volatilities, and at cryogenic temperatures that difference becomes a practical lever for separation and concentration. Modern literature still characterizes Kr/Xe purification as energy-intensive cryogenic distillation in many conventional routes.
Key engineering levers procurement teams should ask about:
Column design (tray/packing selection, reflux ratio targets)
Heat integration (specific heat exchange approach and cold recovery)
Control philosophy for stable cut points and product purity
The final jump from “high purity” to 6N is often achieved using polishing technologies such as:
Getter-based purification (reactive scavengers for trace O₂/H₂O, etc.)
Adsorption polishing (application-specific media)
PSA-style finishing in certain configurations
At this stage, the focus is not bulk separation—it’s trace impurity removal plus verification via trace impurity analysis aligned to the end-use spec.
Noble gases are used across semiconductor process steps, and excimer laser technology relies on noble gas mixtures (often including krypton or xenon, depending on wavelength and system design).
For fabs, purity translates into:
repeatability (less drift from contaminants)
fewer tool-related excursions
more predictable laser gas and process gas performance
Xenon is widely used as a propellant in electric propulsion systems. NASA’s NEXT (Evolutionary Xenon Thruster) program is a well-known example of xenon-based ion propulsion development and documentation.
Hall-effect thrusters also commonly use xenon—and in some designs krypton is also used as a propellant option.
Krypton remains valued for performance in certain lighting and insulated glass applications, where its thermal properties can improve insulation performance compared with air fills.
For many organizations, the “new supply” strategy is less about hoping for more upstream production—and more about:
improving yield from existing ASUs
reducing venting or losses from purge streams
building resilience against supply shocks
This is where rare gas recovery becomes both an engineering upgrade and a sustainability-aligned operational decision.
Selecting a rare gas recovery partner is not just about nameplate purity. It’s about uptime, repeatability, and lifecycle cost—especially when recovery skids must integrate with an operating ASU.
Fortune Gas positions itself as a cryogenic technology provider with rare gas capabilities, focusing on engineered systems and process equipment that operate reliably under extreme low-temperature conditions.
At cryogenic temperatures, seemingly small design choices become failure modes:
thermal stress and fatigue
seal/material compatibility
contaminant freeze-out risks
instrumentation drift and calibration challenges
A robust recovery system design is one that assumes real plant variability—and still holds product specs.
Rare gas projects range from R&D-scale purification loops to full ASU tie-ins. Fortune Gas supports rare gas solutions as part of a broader cryogenic portfolio, enabling practical integration paths for different capacities and site constraints.
As demand increases across semiconductors and space propulsion, rare gas recovery is becoming the most realistic way to stabilize supply, improve yield, and control purity at the 6N level. The winning programs treat recovery as an end-to-end system: feed characterization, front-end cleanup, cryogenic distillation, final polishing, and validated trace analysis.
Rare gas recovery is the process of capturing, purifying, and recycling noble gases (such as krypton and xenon) from industrial streams—commonly oxygen-enriched or purge streams associated with ASUs—and upgrading them into high-purity products suitable for reuse.
6N means 99.9999% purity, implying extremely low total impurities (on the order of ~1 ppm overall), with many applications requiring specific contaminants (like H₂O or O₂) to be controlled at very low levels.
Because Kr and Xe have different volatilities at cryogenic temperatures, distillation can separate and concentrate them effectively—though it is often energy-intensive and typically supported by upstream cleanup and downstream polishing.